Testing Plant Substances as Potential Medicines
Purpose:
What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
- Balance, weigh boat, lab scoops
- LB broth base
- Media bottles, 250 mL
- Sterilizer/autoclave
- Water bath, 37*C, shaking
- Sterile LB agar
- Laminar flow hood and disinfectant
- Plastic safety glasses
- Bunsen burner and gas lighter
- Inoculating loop, Ni/Cr wire
- Petri dishes, 60x15mm, sterile
- E. coli JM109 (stock plate)
- Plant specimen
- Mortar and pestle
- Pipet, 10 mL and pump
- Plastic funnels, short-stemmed
- Filter paper disks, 5mm diameter
- 100 mL beakers
- Syringe, 10 mL and filter, 0.2 micrometers
- Reaction tubes and rack, 1.7 mL
- Methanol, absolute
- Pipet, 1 mL and pump
- Dry block heater/heat block
- Forceps, fine-tipped
- Ampicillin
- Glass spreader
- Incubator oven, 37*c
Part II:
4. Grind 2g plant material in mortar and pestle to break open cells with 10ml di H2O
- Let sit for 3 minutes
- Filter through paper lined funnel
- Filter sterilize 1ml into microtube
5. Repeat step 4 but replace water with methanol extracting solvent, place 1 ml extract into 1.7 ml tube inside 65 degree Celsius heat block, for 1 day to evaporate the methanol, reconstitute in 1ml di H2O
6. For other samples repeat 4 and 5, there should be 6 tubes
7. Use sterile forceps to drop filter papers into each tube of extracts
8. Prepare negative control disks, three each, only methanol and sterile distilled water
9. Prepare 6 positive controls of ampicillin
10. Allow sufficient times for the disks to soak the extracts, perhaps overnight
11. Close tubes, store at 4 degrees Celsius until ready
- Let sit for 3 minutes
- Filter through paper lined funnel
- Filter sterilize 1ml into microtube
5. Repeat step 4 but replace water with methanol extracting solvent, place 1 ml extract into 1.7 ml tube inside 65 degree Celsius heat block, for 1 day to evaporate the methanol, reconstitute in 1ml di H2O
6. For other samples repeat 4 and 5, there should be 6 tubes
7. Use sterile forceps to drop filter papers into each tube of extracts
8. Prepare negative control disks, three each, only methanol and sterile distilled water
9. Prepare 6 positive controls of ampicillin
10. Allow sufficient times for the disks to soak the extracts, perhaps overnight
11. Close tubes, store at 4 degrees Celsius until ready
Part III:
12. Use a clean pipet to move 1 ml of E.coli broth to the middle of a petri dish, sterilize a spreading loop with fire and alcohol, to spread the bacteria evenly. Cover, and leave for at least 15 minutes
13. Using forceps, place 1 dish in each quadrant, 2 cm from the edge of the petri dish, place the methanol samples in one dish and the water samples on another
14. Repeat step 13, in order to end up with 3 methanol and 3 water replicates
15. Place a negative control disk in the center of the appropriate plate, then a positive control with ampicillin in a quadrant on each plate
16. In the end you should have 6 petri dishes with a negative control in the middle and a a positive control and three sample disks
17. Guarantee that the disk hold fast to surface of agar, alter the plates and brood at 37*C for 24 to 48 hours
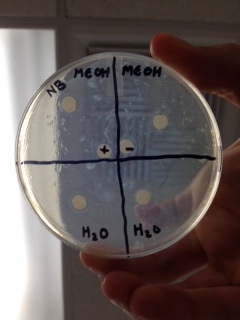
18. After incubation, search for at the plates with plant concentrate circles for zones of inhibition, clear rasnge shaped by inhibitory activity of a substance in the plant material around the circle, photo the plates, marking any restraint of bacterial development
19. Make an information table for the repeats and midpoints, incorporate depictions of the bacterial garden around every circle, record the breath and clarity of any cleared zones around the circles in quantitative estimations
13. Using forceps, place 1 dish in each quadrant, 2 cm from the edge of the petri dish, place the methanol samples in one dish and the water samples on another
14. Repeat step 13, in order to end up with 3 methanol and 3 water replicates
15. Place a negative control disk in the center of the appropriate plate, then a positive control with ampicillin in a quadrant on each plate
16. In the end you should have 6 petri dishes with a negative control in the middle and a a positive control and three sample disks
17. Guarantee that the disk hold fast to surface of agar, alter the plates and brood at 37*C for 24 to 48 hours
18. After incubation, search for at the plates with plant concentrate circles for zones of inhibition, clear rasnge shaped by inhibitory activity of a substance in the plant material around the circle, photo the plates, marking any restraint of bacterial development
19. Make an information table for the repeats and midpoints, incorporate depictions of the bacterial garden around every circle, record the breath and clarity of any cleared zones around the circles in quantitative estimations
Data Analysis/Conclusion:
- Yes one of the H2O extract discs represented a string positive, while one of the methanol extracts and the ampicillin showed a weak positive possibly negative.
- My controls did somewhat work as I expected. To the extent of my vision, I saw that the ampicillin disc represented a weak positive as it should've been a strong positive while the water indicator was a strong negative as it was supposed to.
- Errors that could have possibly explained my results could be that; the ampicillin solution wasn't valid, invalid e coli, human error, putting the wrong disc somewhere it shouldn't have been, using non-sterile instruments, or contaminating the subject.
- Further experimentation could include; an entire retest of the experiment, this time using more reliable ampicillin solutions, or using different e coli, or being more careful with common human error, sterilization techniques.
- From here we would look at our results and analyze what went different and didn't change. New experiments may need to be conducted to conclude a perfect result.
- My controls did somewhat work as I expected. To the extent of my vision, I saw that the ampicillin disc represented a weak positive as it should've been a strong positive while the water indicator was a strong negative as it was supposed to.
- Errors that could have possibly explained my results could be that; the ampicillin solution wasn't valid, invalid e coli, human error, putting the wrong disc somewhere it shouldn't have been, using non-sterile instruments, or contaminating the subject.
- Further experimentation could include; an entire retest of the experiment, this time using more reliable ampicillin solutions, or using different e coli, or being more careful with common human error, sterilization techniques.
- From here we would look at our results and analyze what went different and didn't change. New experiments may need to be conducted to conclude a perfect result.
1. It does mean that it isn't an antimicrobial agent for e coli, however it could be antimicrobial for another type of bacteria. So it may be, but there is a possibility that it could be an antimicrobial agent.
2. Because if it would be testing the alcohol instead.
3. In order to find the extract causing the antimicrobial action, we must isolate the compounds on the microscopic level. Once all the compounds are isolated, they have to be tested individually to determine which compound causes this.
2. Because if it would be testing the alcohol instead.
3. In order to find the extract causing the antimicrobial action, we must isolate the compounds on the microscopic level. Once all the compounds are isolated, they have to be tested individually to determine which compound causes this.